21 CFR 820.198 – Implementing Risk-Based Management in Handling Complaints
April 15th, 2013 // 2:48 pm @ jmpickett
More Exclusive FDA cGMP Compliance News
Complaint Handling – 21 CFR 820.198
A strong complaint-handling process, according to 21 CFR 820.198, is a system of management that guarantees the highest possible level of patient safety. It also secures the highest possible investment return and it reduces product liability lawsuits to the minimum. A good complaint-handling system is a gauge of post market quality that measures and controls business, patient and regulatory risks.
All of the employees in a device company should be able to perform their essential duties according to your company’s SOPs after they have had enough training. Medical device companies should have a unit that is assigned to handle complaints and set up procedures. This group should:
- Define the process of handling complaints
- Document all of the steps to receive, review and go over/evaluate complaints
- Implement the procedures with strong employee training
If the SOPs above are followed, there will be a standardized and complete process of complaint handling. It is vital that a multidisciplinary team is working on the system as the procedures are devised and later approved. Like any other developmental process, the first implementation will require a considerable learning curve. What makes it easier to learn is if your procedures are well written and simple to follow.
Complaint handling is a very integrated process. So, it is ideal to break down this process into a variety of SOPs. Some of the SOP categories for complaints include:
- Customer service/complaint handling
- Complaints and filing of MDRs
- Investigation of complaints
- MDR
- Product recall
- CAPA
- Product repair/service
- Complaint software
We recommend flow charting your entire complaint handling system, and also the creation of decision trees is helpful. Process flow charts are very helpful visual aids for complex complaint handling processes.
May 30 Webinar – Essentials of Complaint Handling and Post Marketing Management
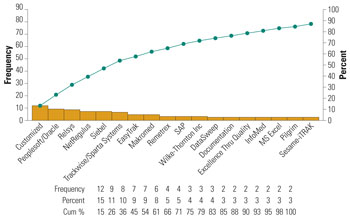
Once you have a good system and corresponding procedures set up, most mishandling and misplacement problems can be quickly solved. It is a good idea to switch from paper to electronic systems to track complaints. This will allow you to have more complete documentation of problems, a better and more coordinated resolution and less turnaround time. There are a number of commercial off the shelf (COTS) software programs that will provide you with a very high tech platform to connect a specific complaint to a specific failure investigation and the corresponding corrective action. The image below provides you with some of the most common commercial software used by device companies to manage risk.
Some of these computer applications will give you trending analysis and other tools for statistical modeling to enhance your risk based complaint handling processes.
For complaints that arrive from many different sources, it is important to learn how these are captured and entered into the system. Software is very useful to do this. Managers should direct complaint handling efforts to the most common channels of communication to capture information. The most common channels that customers use to provide device complaints are:
- Customer call center,
- Company main phone number
- Direct communication with personnel in the field
As the complaint information comes in from customers, call service reps should screen all of these calls and decide if the information is actually a complaint. Then they will assign tracking numbers and enter the information into the complaint handling system. This initial point of decision making is very important. If it is done right, the firm can avoid expensive rework down the road. After this first screening, the QA team should go over the information and approve a complaint initiation process. Everyone in your firm can receive a complaint, so managers should tell all employees to direct these complaints to one processing center, which in this case would be the call servicing center.
The closure of complaints is very dependent upon a good failure investigation. Many device companies often have problems getting enough details about malfunctions or the problem events from the users of the device, the sales team or from clinical people. Also, getting customers to return defective devices is a challenge. If you can’t see the malfunction up close and discover the root cause and analyze risk, it is hard to implement effective remediation.
For more information about effective complaint handling, please refer to our May 30 webinar.
Upcoming Expertbriefings.com Webinars
- May 2 – Avoid the CDRH eCopy Chaos – How to Prepare a Compliant eCopy Submission
- April 25 – How to Survive PREDICT – FDA’s New Import Screening Program
- April 29 – Audit Your Lab Like an Expert FDA Auditor: A Roadmap to Lab Compliance
- May 1 – Avoiding Warning Letter Disasters With a Strong Contractor Quality Agreement
- May 7 – FDA Recall Chief Update – How to Design a Bulletproof Product Recall Strategy
- May 30 – Essentials of Complaint Handling and Post Market Management